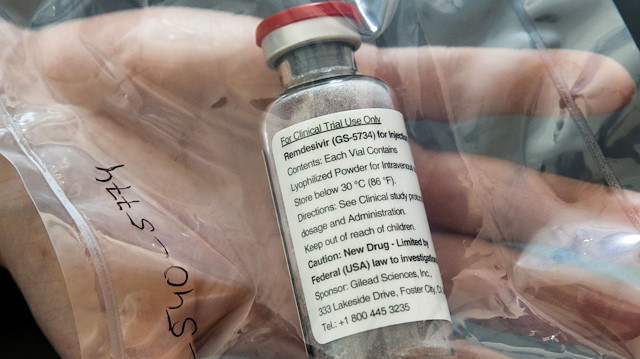
The European Commission on Friday said there had been no change in the authorised uses of Gilead's remdesivir after the World Health Organization advised against using the antiviral for treating hospitalised COVID-19 patients.
"We take note that the WHO has now updated its guidelines on the use of remdesivir," a Commission spokesman said in an email.
The European drugs regulator has requested full data from the WHO-led Solidarity trial into the drug and will assess the evidence, together with other available data, to see if any changes are needed to its market authorisation, he said.